So the molecular weight of Magnesium chloride is 95.2104. Molecular weight of Magnesium chloride (MgCl 2): 24.305 (Magnesium) + 70.90 (Chlorine) = 95.2104 Step 4: Calculate the molecular weight of Magnesium chloride by adding up the total weight of all atoms. So, the number of moles of each compound can be calculated as: moles of MgCl 132.1 g× 1. In this case the density of the water is 1 g/mL. 
Density is the ratio of the weight (mass) of a substance to the volume it occupies. Total weight of chlorine atoms in Magnesium chloride: 35.4527 x 2 = 70.9052 In this case the molar mass of MgCl is 95.21 g/mole and the molar mass of water is 18 g/mole. Number of chlorine atoms in Magnesium chloride: 2 Total weight of magnesium atoms in Magnesium chloride: 24.305 x 1 = 24.305 Number of magnesium atoms in Magnesium chloride: 1 Step 3: Calculate the molecular weight of Magnesium chloride by adding the total weight of all atoms. Step 2: Find out the atomic weights of each atom.Ītomic weight of magnesium (Mg): 24.305 (Ref: Pubchem-12, Jlab-ele012)Ītomic weight of chlorine (Cl): 35.4527 (Ref: Pubchem-Chlorine, jlab-ele017) It is inorganic in nature that may appear as a white or colorless crystalline. MgCl Molar mass, 95.211 g/mol (anhydrous) 203.31 g/mol (hexahydrate) Appearance, white or colourless crystalline solid. The chemical formula shows that one molecule of Magnesium chloride consists of one Magnesium (Mg) atom and two Chlorine (Cl) atoms. MgCl2 is an ionic halide salt consisting of magnesium and chlorine elements. How many grams of MgCl2 power wouldneed to be weighted out to makw200ml of a. To convert this to grams, use the compounds molar mass 9.0moles MgCl2 95.211 g 1mole MgCl2 860 g The answer is rounded to two sig figs, the number of sig figs you have for your values. 
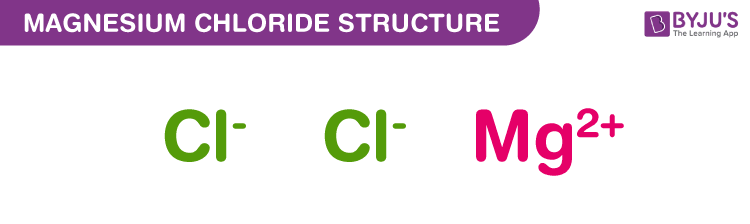
The chemical formula of Magnesium chloride is MgCl 2. Magnesium chloride (MgCl ) has a relative molecular weight of 95.21 g/mol. How many moles of CaCl, are in 250 mL of a 3.0 M of CaCl, solution The O-H bond in water is polar because What is the concentration, in mass percent (m/m), of a solution prepared from 50.0 g. You will know different atoms and their number in a Magnesium chloride molecule from the chemical formula. What is the molar mass of copper Question: Calculate the molar mass of magnesium chloride, MgCl2 Calculate the molar mass of potassium chloride, KCI. Step 1: Find out the chemical formula and determine constituent atoms and their number in a Magnesium chloride molecule. CALCULATION PROCEDURE: Magnesium Chloride (MgCl2) Molecular Weight Calculation The molecular weight of Magnesium chloride is 95.2104 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements. Magnesium chloride (MgCl 2) is an organic compound of two elements: Magnesium and Chlorine. The molecular weight of Magnesium Chloride (MgCl 2) is 95.2104. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
